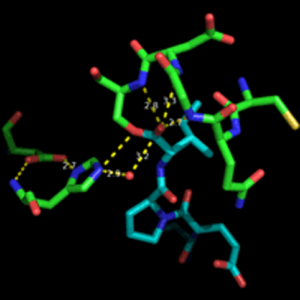
Enzyme kinetics is a challenging concept for students to visualize. Substrate binding to the active site of an enzyme involves multiple weak interactions between the R-groups of the amino acids and the substrate. These weak interactions increase the binding energy, lower the energy of activation, and prevents side reactions. The mechanism used by enzymes to catalyze reactions are complex and involve interactions among the substrates, enzymes, and products. Students will be able to examine the structure of enzymes, substrate binding, substrate specificity, product release, and regeneration of the enzyme active site.
Learning Objectives
- Students will be able to explain how structure determines enzyme specificity.
- Students will be able to predict substrate structural features that are important for substrate binding and catalysis.

